Special-purpose
batteries
Back in the early days of electrical
measurement technology, a special type of battery known as a
mercury standard cell was popularly used as a voltage
calibration standard. The output of a mercury cell was
1.0183 to 1.0194 volts DC (depending on the specific design
of cell), and was extremely stable over time. Advertised
drift was around 0.004 percent of rated voltage per year.
Mercury standard cells were sometimes known as Weston
cells or cadmium cells.
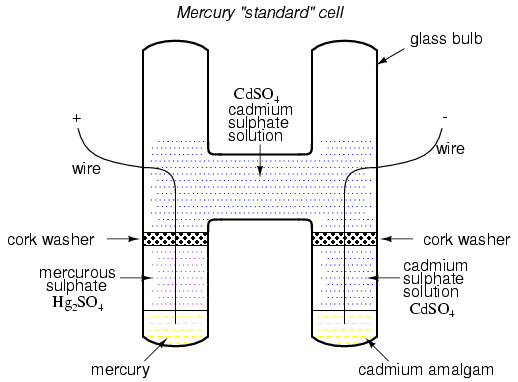
Unfortunately, mercury cells were rather
intolerant of any current drain and could not even be
measured with an analog voltmeter without compromising
accuracy. Manufacturers typically called for no more than
0.1 mA of current through the cell, and even that figure was
considered a momentary, or surge maximum!
Consequently, standard cells could only be measured with a
potentiometric (null-balance) device where current drain is
almost zero. Short-circuiting a mercury cell was prohibited,
and once short-circuited, the cell could never be relied
upon again as a standard device.
Mercury standard cells were also susceptible
to slight changes in voltage if physically or thermally
disturbed. Two different types of mercury standard cells
were developed for different calibration purposes:
saturated and unsaturated. Saturated standard
cells provided the greatest voltage stability over time, at
the expense of thermal instability. In other words, their
voltage drifted very little with the passage of time (just a
few microvolts over the span of a decade!), but tended to
vary with changes in temperature (tens of microvolts per
degree Celsius). These cells functioned best in
temperature-controlled laboratory environments where
long-term stability is paramount. Unsaturated cells provided
thermal stability at the expense of stability over time, the
voltage remaining virtually constant with changes in
temperature but decreasing steadily by about 100 �V every
year. These cells functioned best as "field" calibration
devices where ambient temperature is not precisely
controlled. Nominal voltage for a saturated cell was 1.0186
volts, and 1.019 volts for an unsaturated cell.
Modern semiconductor voltage (zener diode
regulator) references have superseded standard cell
batteries as laboratory and field voltage standards.
A fascinating device closely related to
primary-cell batteries is the fuel cell, so-called
because it harnesses the chemical reaction of combustion to
generate an electric current. The process of chemical
oxidation (oxygen ionically bonding with other elements) is
capable of producing an electron flow between two electrodes
just as well as any combination of metals and electrolytes.
A fuel cell can be thought of as a battery with an
externally supplied chemical energy source.
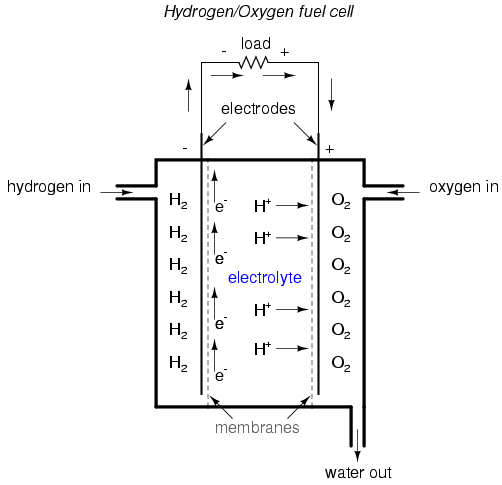
To date, the most successful fuel cells
constructed are those which run on hydrogen and oxygen,
although much research has been done on cells using
hydrocarbon fuels. While "burning" hydrogen, a fuel cell's
only waste byproducts are water and a small amount of heat.
When operating on carbon-containing fuels, carbon dioxide is
also released as a byproduct. Because the operating
temperature of modern fuel cells is far below that of normal
combustion, no oxides of nitrogen (NOx) are
formed, making it far less polluting, all other factors
being equal.
The efficiency of energy conversion in a
fuel cell from chemical to electrical far exceeds the
theoretical Carnot efficiency limit of any
internal-combustion engine, which is an exciting prospect
for power generation and hybrid electric automobiles.
Another type of "battery" is the solar
cell, a by-product of the semiconductor revolution in
electronics. The photoelectric effect, whereby
electrons are dislodged from atoms under the influence of
light, has been known in physics for many decades, but it
has only been with recent advances in semiconductor
technology that a device existed capable of harnessing this
effect to any practical degree. Conversion efficiencies for
silicon solar cells are still quite low, but their benefits
as power sources are legion: no moving parts, no noise, no
waste products or pollution (aside from the manufacture of
solar cells, which is still a fairly "dirty" industry), and
indefinite life.
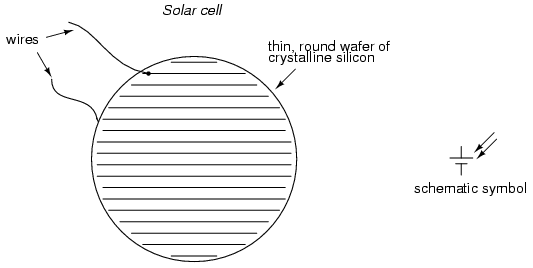
Specific cost of solar cell technology
(dollars per kilowatt) is still very high, with little
prospect of significant decrease barring some kind of
revolutionary advance in technology. Unlike electronic
components made from semiconductor material, which can be
made smaller and smaller with less scrap as a result of
better quality control, a single solar cell still takes the
same amount of ultra-pure silicon to make as it did thirty
years ago. Superior quality control fails to yield the same
production gain seen in the manufacture of chips and
transistors (where isolated specks of impurity can ruin many
microscopic circuits on one wafer of silicon). The same
number of impure inclusions does little to impact the
overall efficiency of a 3-inch solar cell.
Yet another type of special-purpose
"battery" is the chemical detection cell. Simply put,
these cells chemically react with specific substances in the
air to create a voltage directly proportional to the
concentration of that substance. A common application for a
chemical detection cell is in the detection and measurement
of oxygen concentration. Many portable oxygen analyzers have
been designed around these small cells. Cell chemistry must
be designed to match the specific substance(s) to be
detected, and the cells do tend to "wear out," as their
electrode materials deplete or become contaminated with use.
-
REVIEW:
-
mercury standard cells are special
types of batteries which were once used as voltage
calibration standards before the advent of precision
semiconductor reference devices.
-
A fuel cell is a kind of battery
that uses a combustible fuel and oxidizer as reactants to
generate electricity. They are promising sources of
electrical power in the future, "burning" fuels with very
low emissions.
-
A solar cell uses ambient light
energy to motivate electrons from electrode to another,
producing voltage (and current, providing an external
circuit).
-
A chemical detection cell is a
special type of voltaic cell which produces voltage
proportional to the concentration of an applied substance
(usually a specific gas in ambient air).
|